Diversity in Ophthalmic Clinical Trials
The lack of diversity in clinical trials undermines the generalizability, quality, and external validity of medical research.1 Traditionally, clinical trial recruitment has employed rationales of limited heterogenicity and interindividual variability to establish consistent short-term results; however, diversity and inclusion in ophthalmic clinical trials serve several purposes.2,3
- It gives researchers more information on how new treatments may affect patients of different colors, ethnicities, genders, backgrounds.3
- It provides access to treatments to a wider range of people. These treatments have the potential to improve vision or reduce or prevent eye disease.3
- Participation in the trials from diverse groups gives broader data on treatment outcomes.3
- More diverse clinical trial participation helps reduce care disparities within ophthalmology.3
An analysis of participation in clinical trials from around the globe completed by the US Food and Drug Administration (FDA) in 2020 found that among nearly 292,537 participants, 76% were White, underrepresenting the diversity of the world, with authors stating “Data for global populations are lacking, and current guidelines and clinical decisions are based on insufficiently diverse trials and studies.” 2,4
A similar analysis of ClinicalTrials.gov trial enrollees from 2000 to 2020 found that 79.7% of participants were White and 10% were Black, with much smaller percentages for other minority groups.5 Of the 20,692 trials in the analysis, only 43% reported race and ethnicity data.5
Within ophthalmology specifically, the lack of diversity within clinical trials also appears to be an overarching trend.
- Only 43.6% of all ophthalmology clinical trials reported any racial, ethnic, or sex demographic information of study participants over the last two decades (1997 to 2022).6
- Among 8003 participants across 19 anti-VEGF RCTs for age-related macular degeneration (AMD), 92.3% were Caucasian; Asian, African American, Hispanic/Latino, and American Indian participants collectively comprised just over 5%.7
- Black patients were underrepresented by a 3.0 disparity and by a 4.5-fold disparity in an analysis of 25 National Institutes of Health and industry-sponsored trials of diabetic retinopathy (DR) or macular edema. Among DR trials, researchers found a 2.1-fold disparity compared with disease burden.8
- Hispanic participants were most frequently underrepresented in 23 DME and retinal vein occlusion randomized controlled trials comparing racial/ethnic demographics of trial participants to 2010 Census Bureau results.8 “The representation gap for Hispanic/Latino participants is particularly concerning, given the burden of diabetes and hypertension in this community and the fact that this is the fastest-growing demographic subset in the US,” writes Dr Sally Baxter.8,9
- Black, Hispanic or Latino, and other non-White participants were underrepresented in clinical trials leading to FDA new molecular entity drug approvals in AMD, DR, and open-angle glaucoma in a cohort study examining 20 years of data. While there was some improvement in representation between 2000 and 2020, the researchers concluded that more diversity is needed. If the current trends remain, there could be a continued overrepresentation of White participants by 2050, particularly in AMD trials.10
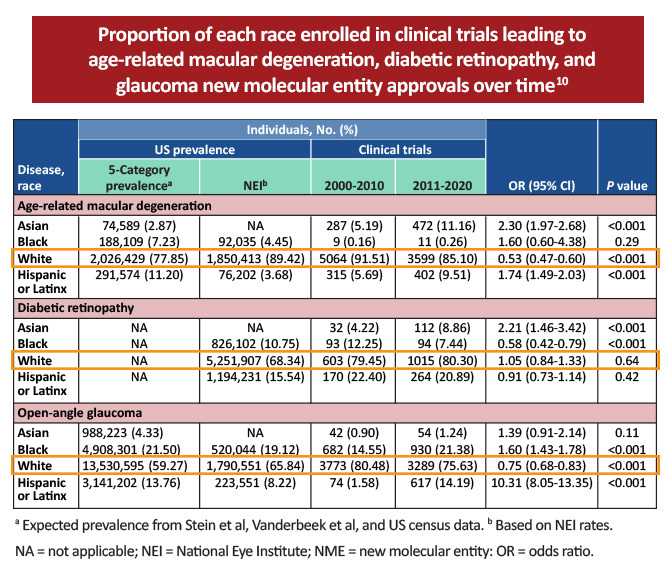
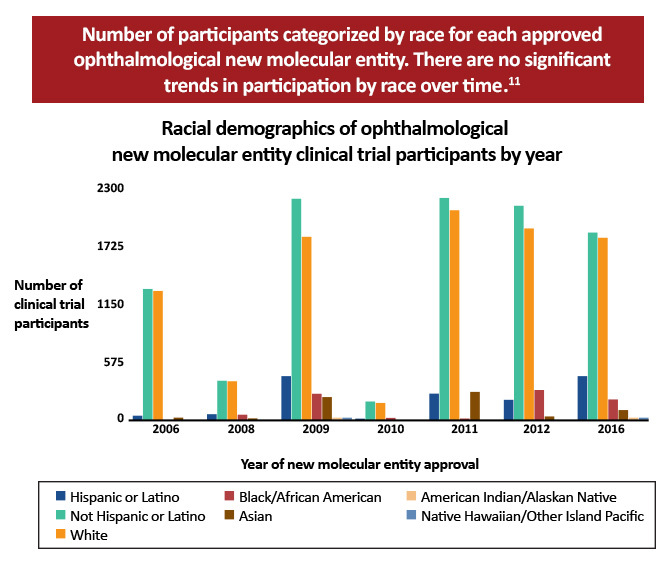
- A study reviewing trials supportive of nine approved ophthalmological new molecular entities from 2006-2016 across a variety of eye diseases revealed significant enrollment variation across the trials, with 1%-15% Black participation and 2%-18% Hispanic participation.10
- Despite the fact that primary open-angle glaucoma is most likely to affect Black individuals, racial and ethnic minority groups had a low participation rate in primary open-angle glaucoma clinical trials, according to a meta-analysis of clinical trials taking place from 1994 to 2019.12 Among the 105 trials with a total 33,428 participants, 70.7% were White, 16.8% were Black, 3.4% were Hispanic/Latino, and 9.1% were from other races/ethnicities.12
- Geographical access is a barrier to trial access and participant diversity. Outside of the Northeastern United States, there was a higher travel burden associated with clinical trial sites for diabetic eye disease, according to researchers in a cross-sectional, retrospective study published in 2021.13
Factors affecting clinical trial participation
There may be several reasons why clinical trials within ophthalmology and medicine in general have had more underrepresentation:9,13
- Less recruitment among underrepresented minorities
- Linguistic barriers
- Cultural barriers
- Structural distrust toward the health system
- Difficulties accessing health services
- Geographic barriers
Improving diversity in clinical trials: implications for ophthalmologists
Increasing diversity within ophthalmic clinical trials, including those within the retina subspecialty, has various underlying factors that need to be addressed to foster trust, enhance awareness, and facilitate participation. Efforts on the individual and collective level can do several things to improve diversity within clinical trials.
On the collective level, the American Academy of Ophthalmology (AAO) has established a task force focused on health equity in eye care. This task force takes a closer look at topics, such as the social determinants of health and medical care, access to care, and care outcomes. The AAO is also committed to developing meaningful patient and provider resources to help address this need for greater diversity, equity, and inclusion.14
Additionally, both pharmaceutical companies and the FDA are making efforts to focus on inclusion within clinical trials for all specialties. In its’ December 2025 “Enhancing Participation in Clinical Trials — Eligibility Criteria, Enrollment Practices, and Trial Design” document, the FDA suggested several ways that clinical trials can increase diversity and inclusion.15 Some of its recommendations, along with recommendations from other sources, include:15,16
- Avoiding unnecessary exclusions and broadening criteria to better reflect the real-world population that will use the drug if approved.
- Improving enrollment practices to make clinical trials less burdensome and more accessible to a wider variety of participants.
- Using innovative trial designs, such as adaptive, decentralized, or enrichment strategies.
- Using more inclusive strategies for trial outreach.
- Working with community members on trial design to pinpoint where trials may discourage participation, so changes can be made to modify those areas.
- Using clinical trial sites located in racially and ethnically diverse areas.
- Holding frequent recruitment events, including evenings and weekends.
- Considering the use of online and social media recruitment strategies.
- Involving anthropologists to help reach communities with systemic distrust due to previous health-related injustice.4
- Recruiting a more diverse staff; research from Bains et al. found that trial participation increased when patients were approached by an ophthalmic staff member of the same race or ethnicity.17
- Enhancing health literacy and encouraging trial participation for by offering educational and trial resources into native languages of community patients.
Although some studies have shown some narrowing of disparities and racial/ethnicity reporting in ophthalmic trial underrepresentation, continued efforts for engagement with underrepresented groups to diversify trial composition are vital to ensure the quality and validity of trial results.7,8,18,19 Programs, such as the National Institute of Health’s All of Us bring focused efforts to increase diversity in clinical trials.9,20
References
- Collister D, Song C, Ruzycki SM. Fostering diversity in clinical trials: need for evidence and implementation to improve representation. BMJ Med. 2024;3. https://bmjmedicine.bmj.com/content/3/1/e000984
- Sharma A, Palaniappan L. Improving diversity in medical research. Nat Rev Dis Primers. 2021;7:74. https://www.nature.com/articles/s41572-021-00316-8
- Schwartz AL, Alsan M, Morris AA, et al. Why diverse clinical trial participation matters. N Engl J Med. 2023;388:1252-1254. https://www.nejm.org/doi/full/10.1056/NEJMp2215609
- Sanjiv N, Osathanugrah P, Harrell M, et al. Race and ethnic representation among clinical trials for diabetic retinopathy and diabetic macular edema within the United States: A review. J Natl Med Assoc. 2022;114:123-140. https://www.sciencedirect.com/science/article/abs/pii/S0027968421002340
- Turner BE, Steinberg JR, Weeks BT, Rodriguez F, Cullen MR. Race/ethnicity reporting and representation in US clinical trials: A cohort study. Lancet Reg Health Am. 2022;11:100252. https://www.sciencedirect.com/science/article/pii/S2667193X22000692
- Shafiee A, Juran T, Zabaneh I, et al. Racial and gender disparities in clinical trial representation for age-related macular degeneration treatments: a scoping review. J Clin Transl Ophthalmol. 2025;3:16. https://www.mdpi.com/2813-1053/3/3/16
- Kaakour AH, Hua HU, Rachitskaya A. Representation of race and ethnicity in randomized clinical trials of diabetic macular edema and retinal vein occlusion compared to 2010 US Census data. JAMA Ophthalmol. 2022;140:1096-1102. https://jamanetwork.com/journals/jamaophthalmology/fullarticle/2797004
- Baxter SL. Representation matters—Diversity in clinical trials. JAMA Ophthalmol. 2022;140:1103‑1104. https://jamanetwork.com/journals/jamaophthalmology/article-abstract/2797007
- Berkowitz ST, Groth SL, Gangaputra S, Patel S. Racial/ethnic disparities in ophthalmology clinical trials resulting in US Food and Drug Administration clinical trials from 2000 to 2020. JAMA Ophthalmol. 2021;139:629-637. https://jamanetwork.com/journals/jamaophthalmology/fullarticle/2778782
- Birnbaum FA. Gender and ethnicity of enrolled participants in US Food and Drug Administration (FDA) clinical trials for approved ophthalmological new molecular entities. J Natl Med Assoc. 2018;110:473-479. https://www.sciencedirect.com/science/article/abs/pii/S0027968416302036
- Allison K, Patel DG, Greene L. Racial and ethnic disparities in primary open-angle glaucoma clinical trials: A systemic review and meta-analysis. JAMA Netw Open. 2021;4:e218348. https://jamanetwork.com/journals/jamanetworkopen/fullarticle/2779939
- Soares RR, Parikh D, Shields CN, et al. Geographic access disparities to clinical trials in diabetic eye disease in the United States. Ophthalmol Retina. 2021;5:879-887. https://www.sciencedirect.com/science/article/abs/pii/S2468653020304875
- American Academy of Ophthalmology. Meeting the needs of all our patients. https://www.aao.org/meeting-needs-of-all-patients
- US Food and Drug Administration. Enhancing participation in clinical trials — eligibility criteria, enrollment practices, and trial designs. Published December 15, 2025. https://www.fda.gov/regulatory-information/search-fda-guidance-documents/enhancing-participation-clinical-trials-eligibility-criteria-enrollment-practices-and-trial-designs
- Coney JM. Racial bias in clinical trials: What you need to know. Retina Today. March 2021. https://retinatoday.com/articles/2021-mar/racial-bias-in-clinical-trials-what-you-need-to-know
- Bains A, Osathanugrah P, Sanjiv N, et al. Diverse research teams and underrepresented groups in clinical studies. JAMA Ophthalmol. 2023;141:1037-1044. https://jamanetwork.com/journals/jamaophthalmology/article-abstract/2810430
- Goodman D, Prasad M, Sanjiv N, et al. Enrollment of race/ethnicity in ophthalmic clinical trials before and after the Final Rule. J Natl Med Assoc. 2025;117:285-292. https://www.sciencedirect.com/science/article/pii/S0027968425000768
- Chang SJ, Grewal KS, Selvam A, et al. Ethnoracial representation in united states clinical trials for retinal vein occlusion. Am J Ophthalmol. 2025;276:186-194. https://www.ajo.com/article/S0002-9394(25)00183-7/fulltext
- All of Us Research Program. National Institutes of Health. Published 2025. https://allofus.nih.gov/
All URLs accessed January 12, 2025.

