This activity is provided by Med Learning Group.
This activity is supported by an independent medical education grant from Regeneron Pharmaceuticals, Inc.
Copyright © 2019Med Learning Group. Built by Divigner. All Rights Reserved.
Traditionally, clinical trial recruitment has employed rationales of limited heterogenicity and interindividual variability to establish consistent short-term results; however, diversity and inclusion in ophthalmic clinical trials serve several purposes1:
An analysis of participation in clinical trials from around the globe completed by the US Food and Drug Administration (FDA) in 2020 found that among nearly 292,537 participants, 76% were White, underrepresenting the diversity of the world.1 “Data for global populations are lacking, and current guidelines and clinical decisions are based on insufficiently diverse trials and studies,” according to A. Sharma and L. Palaniappan.1
A similar analysis of ClinicalTrials.gov trial enrollees from 2000 to 2020 found that 79.7% of participants were White and 10% were Black, with much smaller percentages for other minority groups.3 Of the 20,692 trials in the analysis, only 43% reported race and ethnicity data.3
Within ophthalmology specifically, the lack of diversity within clinical trials also appears to be an overarching trend, based on the following studies:
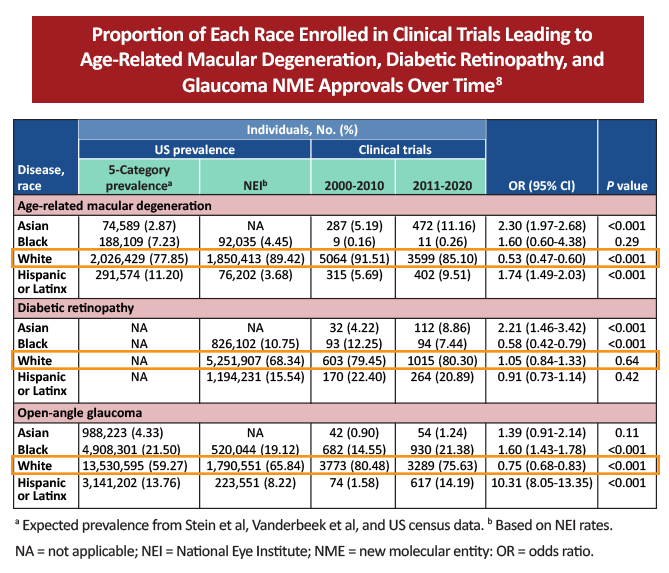
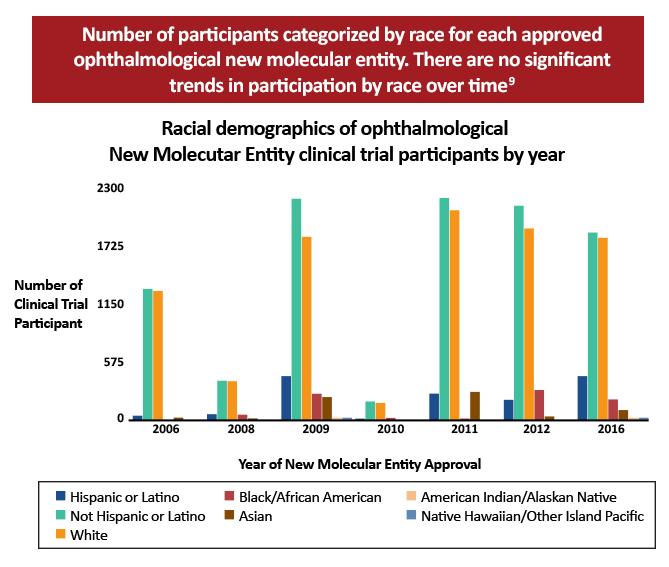
There may be several reasons why clinical trials within ophthalmology and medicine in general have had more underrepresentation:6
Increasing diversity within ophthalmic clinical trials, including those within the retina subspecialty, has various underlying factors that need to be addressed to foster trust, enhance awareness, and facilitate participation. Efforts on the individual and collective level can do several things to improve diversity within clinical trials.
On the collective level, the American Academy of Ophthalmology (AAO) has established a task force focused on health equity in eye care. This task force takes a closer look at topics, such as the social determinants of health and medical care, access to care, and care outcomes. The AAO is also committed to developing meaningful patient and provider resources to help address this need for greater diversity, equity, and inclusion.12
Additionally, both pharmaceutical companies and the FDA are making efforts to focus on inclusion within clinical trials for all specialties. In its November 2020 “Enhancing the Diversity of Clinical Trial Populations” document, the FDA suggested several ways that clinical trials can increase diversity and inclusion.13 Some of its recommendations, along with recommendations from other sources, include:
Although studies such as Yu et al.16 have shown some narrowing of disparities and racial/ethnicity reporting in ophthalmic trial underrepresentation, continued efforts for engagement with underrepresented groups to diversify trial composition are vital to ensure the quality and validity of trial results.5 Programs, such as the National Institute of Health’s All of Us bring focused efforts to increase diversity in clinical trials.6
All URLs accessed February 21, 2024
This activity is provided by Med Learning Group.
This activity is supported by an independent medical education grant from Regeneron Pharmaceuticals, Inc.
Copyright © 2019Med Learning Group. Built by Divigner. All Rights Reserved.
